
The trial will be evaluating AXPAXLI dosed every 6 months versus aflibercept (2 mg) dosed every 8 weeks in treatment-naïve wet AMD patients.

The trial will be evaluating AXPAXLI dosed every 6 months versus aflibercept (2 mg) dosed every 8 weeks in treatment-naïve wet AMD patients.

Under the terms of the agreement, 4DMT will grant Otsuka exclusive rights to develop and commercialize 4D-150 for retinal vascular diseases in Japan, China, Australia, and other Asia-Pacific markets.

The funding will support the completion of the clinical PoC of its AAVB-039 CELESTE study and the completion of the STELLA natural history study

Veligrotug is an intravenously delivered, anti-insulin-like growth factor-1 receptor (IGF-1R) antibody for the treatment of thyroid eye disease (TED).

Tsu Shan Chambers, optometrist and filmmaker, blends clinical insight and storytelling to highlight preventable vision loss, retinitis pigmentosa, and the power of ability over disability in her socially conscious film.
To celebrate Ophthalmology Times' 50th anniversary, we asked leading experts what the practice would look like today had optical coherence tomography (OCT), one of the biggest innovations in the field, never been invented.

Clinicians discuss how the treatment compares with epi-off cross-linking, what may shape adoption, and how patient outcomes could improve.

Marguerite B. McDonald, MD, FACS, reflects on a first-year residency case that taught her the importance of trusting her instincts and advocating for patient safety.
Dr. James Chelnis explores innovative energy-based technologies in aesthetic facial surgery, emphasizing safety, personalized treatments, and future advancements in patient care.

Ophthalmologists discuss topography-guided techniques, long-term corneal health, and emerging technologies set to transform refractive surgery.

Specialists discuss diagnostic technologies, treatment strategies, and research that are improving outcomes for young patients

Deborah A. Ferrington, PhD, highlights how breakthroughs in imaging, AI, and stem cell research are reshaping ophthalmology.

To celebrate Ophthalmology Times' 50th anniversary, we asked leading experts what the practice would look like today had optical coherence tomography (OCT), one of the biggest innovations in the field, never been invented.

Deborah Ristvedt, DO, recalls a retinal detachment repair that took an unexpected turn, teaching lasting lessons in patient awareness and surgical presence.

Emerging devices, wearable technology, and home-monitoring tools are reshaping how clinicians diagnose, treat, and manage patients.
NYRVANA is the first-in-human trial and is an open-label, multicenter, dose-escalation study investigating the safety, tolerability, and preliminary efficacy of a single intravitreal injection of SPVN20 over 6 months.

This acquisition will include Adverum’s lead candidate, Ixo-vec, a gene therapy treatment for wet AMD.
The results presented at 2025 European Society of Medical Oncology meeting support neoadjuvant darovasertib as a potential treatment to delay or prevent progression of the disease.

Emerging anti-VEGF agents offer enhanced durability and anatomic outcomes in retinal disease.

To celebrate Ophthalmology Times' 50th anniversary, we asked leading experts what the practice would look like today had optical coherence tomography (OCT), one of the biggest innovations in the field, never been invented.

Peter J. McDonnell, MD, reflects on his early experience managing bullous keratopathy with penetrating keratoplasty and how advances like DMEK have transformed outcomes across corneal, refractive, and cataract surgery.

Regeneron faces another FDA setback for Eylea HD due to manufacturing issues, but plans for new facilities signal future production improvements.

Surgeons weigh in on emerging surgical technologies, premium lens trends, and strategies to optimize patient satisfaction and safety.

Topcon Healthcare enhances its AI capabilities by acquiring Toku, Inc., aiming to revolutionize eye care with advanced predictive health insights.
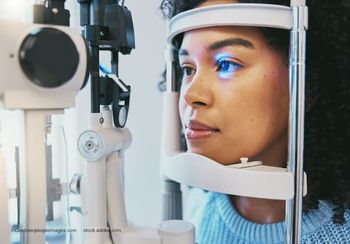
From MIGS integration to remote monitoring and next-generation therapeutics, glaucoma specialists explore how new approaches are redefining patient outcomes.


The AAO 2025 meeting offered a platform for companies to showcase their latest technologies to advance ophthalmic patient care.

Automated standardized graphs facilitate refractive surgery outcome analysis.

To celebrate Ophthalmology Times' 50th anniversary, we asked leading experts what the practice would look like today had optical coherence tomography (OCT), one of the biggest innovations in the field, never been invented.

Oluwatosin U. Smith, MD, reflects on early experiences caring for pediatric patients with complex glaucoma and how evolving tools and advancing science have reshaped treatment options over the decades.